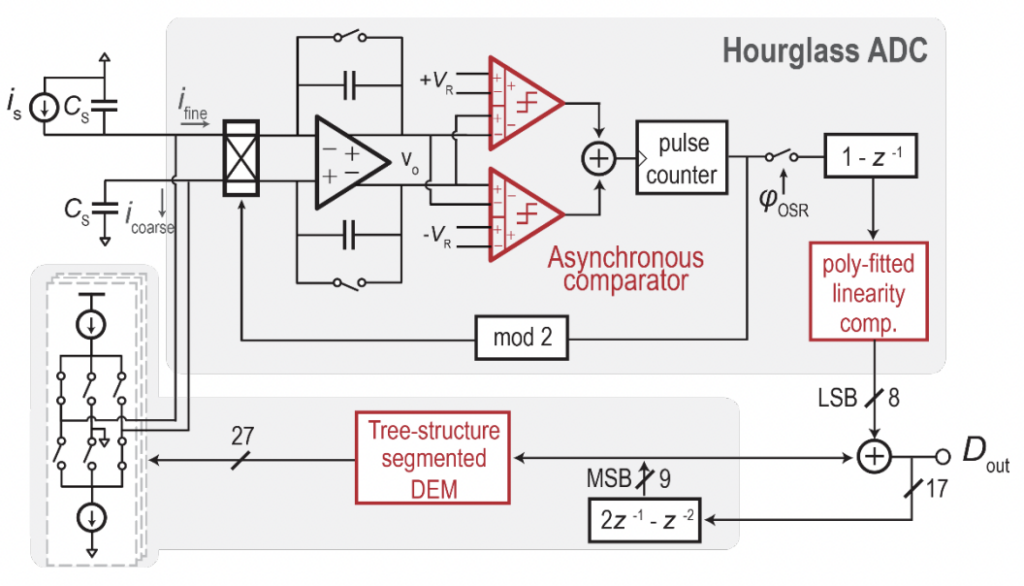
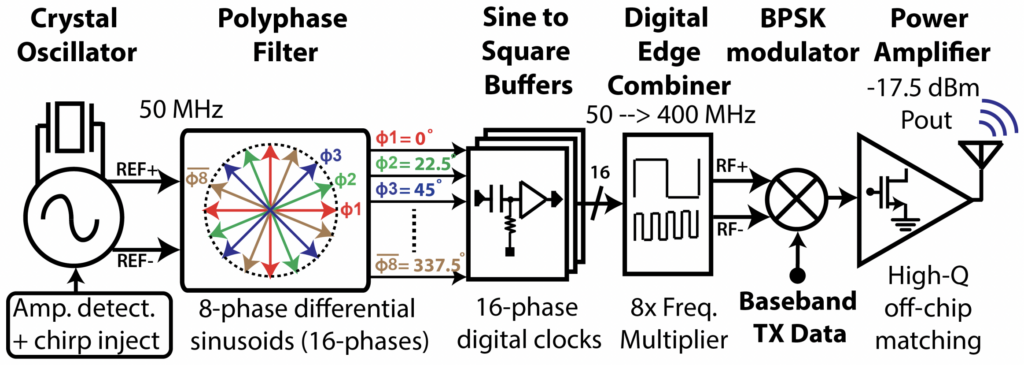
Overview. The BioEE group at the University of California, San Diego (UCSD), led by Prof. Drew Hall, is a multidisciplinary research group performing innovative research in the field of biosensors and bioelectronics. The group uses CMOS circuit design and nanotechnology to develop novel medical devices and high-precision bioinstrumentation.
Why CMOS? Just as the miniaturization of computers, which once filled large rooms, into the microprocessor revolutionized the computer industry, the miniaturization of medical diagnostic tools has the potential to restructure our healthcare system similarly. By moving away from costly and complex biomolecular detection platforms that are constrained to centralized laboratories, one can design cost-effective medical diagnostic tools and permit individuals to take an active part in their health care.
Some examples of current and past research efforts include: in vitro diagnostics, electrochemical biosensors, magnetic biosensors, sensor front-ends, data converters, and wireless communication.
in vitro Diagnostics
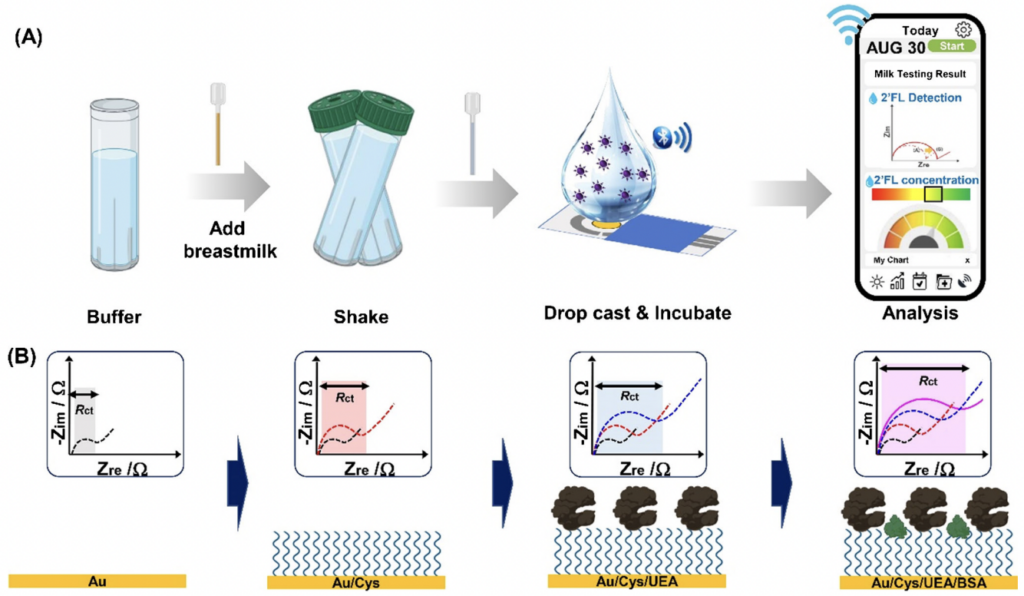
Electrochemical Biosensors
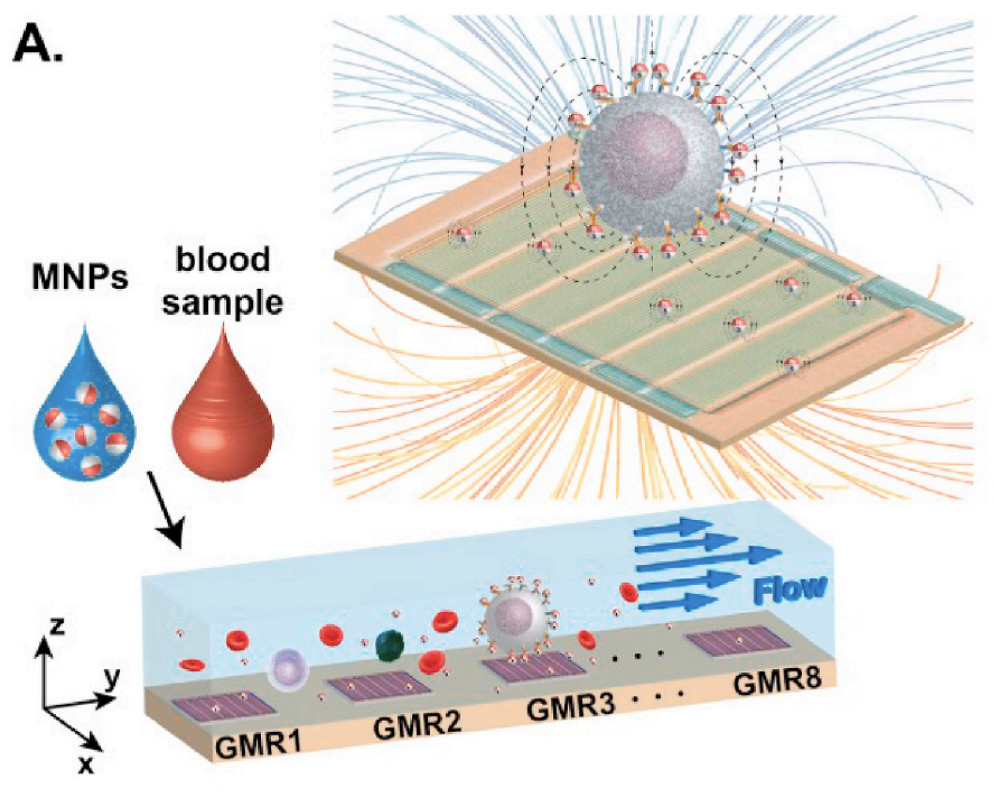
Magnetic Biosensors
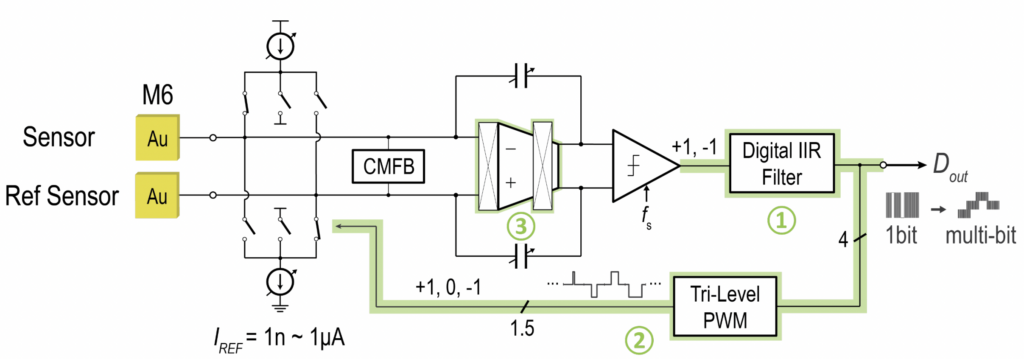
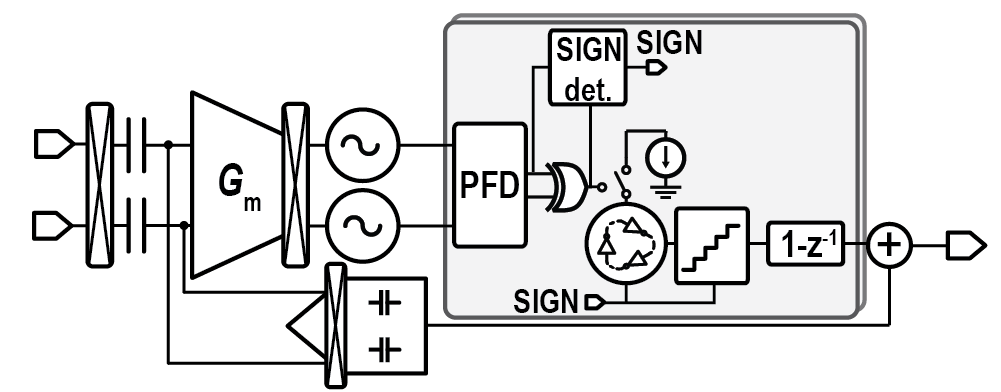
Sensor Front-Ends